  
In 1911, Niels Bohr earned his PhD in Denmark with a dissertation on the electron theory of metals. Bohr’s model became the basis for the quantum theory. Photo: Niels Bohr's research notes for his new atomic theory. An important contribution to the problem of a consistent application of these. In Bohr’s model of the atom each energy level can hold a certain number of electrons. Since he observed only certain colours Bohr suggested that an atom only has certain specific energy levels. It is a result of the combination of the contributions of several scientists, including Max Planck, Albert Einstein, Niels Bohr, Werner Heisenberg, Erwin Schrödinger, and others. The colours of light emitted by the excited hydrogen atoms correspond to the changes of energy that the electrons experience as they move between energy levels. Modern Atomic Theory, also known as quantum mechanics, is the current scientific theory that describes the behavior of atoms and subatomic particles. By applying Michael Plancks quantum physics to the Rutherford structure, Bohr developed his renowned Bohr theory of the atom. As they jump from higher energy levels back down to lower energy levels they emit light energy. Electrons can jump from and to different energy levels within an atom. In 1913, Danish physicist Niels Bohr applied Max Planck’s quantum theory to the nuclear atom of Ernest Rutherford, thus formulating the well-known planetary model of the atom, wherein electrons orbit a central nucleus in well-defined levels of energy ().Note that Bohr stated that electrons in the atom follow elliptical orbits (not circles as is often pictured). As a result electron orbits are sometimes called energy levels. Rutherford had shown that the atom consisted of a. Each orbit has a specific quantity of energy associated with it. In 1913, Bohr published a theory about the structure of the atom based on an earlier theory of Rutherfords. In the planetary model electrons only exist in certain allowed orbits. He realized that hydrogen has a unique atomic spectrum: a pattern of coloured lights that is not produced by any other element. He observed lines of only certain colours of light. h E ( 1 n l o w 2 1 n h i g h 2) 13.6 eV. Bohr explained the hydrogen spectrum in terms of electrons absorbing and emitting photons to change energy levels, where the photon energy is. Bohr directed the light through a prism with a screen behind it. Bohr's model calculated the following energies for an electron in the shell, n. He observed that the hydrogen atoms emitted light when they were “excited” by the additional energy. Understanding Bohr's model requires some knowledge of electromagnetic radiation (or light).īohr's key idea in his model of the atom is that electrons occupy definite orbitals that require the electron to have a specific amount of energy.Bohr also experimented with applying electricity and thermal energy to hydrogen gas. In 1913, the Danish physicist Niels Bohr proposed a model of the electron cloud of an atom in which electrons orbit the nucleus and were able to produce atomic spectra. These difficulties cast a shadow on the planetary model and indicated that, eventually, it would have to be replaced. 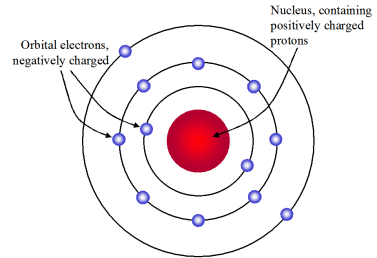
He worked on the Manhattan Project. Furthermore, Rutherford's model was unable to describe how electrons give off light forming each element's unique atomic spectrum. Niels Bohr (7 October 1885 18 November 1962) was a Danish physicist who helped discover quantum physics, the structure of the atom, and the atomic bomb.Bohr was awarded the Nobel Prize in 1922 for, discovering the quantization of atomic energy levels. .jpg)
If the electron circling the nucleus in an atom loses energy, it would necessarily have to move closer to the nucleus as it loses energy, and would eventually crash into the nucleus. Thomson's greatest contribution to science to be his role as a teacher. Thomson published an important monograph in 1913 urging the use of the mass spectrograph in chemical analysis. His atomic theory helped explain atomic bonding and the structure of molecules. This is, after all, how we produce TV signals. Thomson was closely aligned with chemists of the time. It was already known that when a charged particle (such as an electron) moves in a curved path, it gives off some form of light and loses energy in doing so. Unfortunately, there was a serious flaw in the planetary model. \): Niels Bohr with Albert Einstein at Paul Ehrenfest's home in Leiden (December 1925). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
